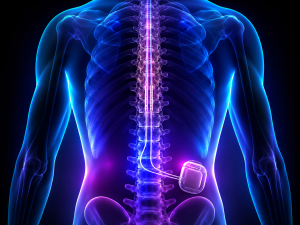
The Bard PowerPort, also called a BardPort, is a port catheter device implanted just under the skin to allow easy attachment to a catheter for intravenous delivery of fluids or medications.
The Bard PowerPort has inherent design and manufacturing flaws that make the device prone to fracturing and migrating out of position. This can cause severe injuries, including internal vascular damage, blood clots, infection, sepsis, pulmonary embolism, organ injury, and the need for surgery to remove or repair the device.
Individuals who suffered injuries because of a defective Bard PowerPort device are now bringing product liability lawsuits against the manufacturer of the PowerPort implants. Our firm is currently accepting new cases from anyone who had a Bard PowerPort port catheter device implanted and was injured because of a fracture, migration, infection, thrombosis, or another implant failure.